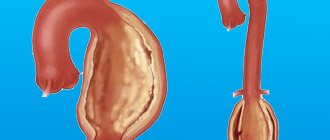
It branches like a tree, first into large branches (trunks), then into smaller branches and twigs, and is conventionally divided into several parts or sections:
- 1. The ascending aorta is the area from the aortic valve to the brachiocephalic trunk.
- 2. The aortic arch is a short section from which all the vessels supplying the arms and head (brachiocephalic arteries) depart. They anatomically form an arch connecting the ascending and descending aorta.
- 3. The descending (thoracic) aorta begins from the mouth of the left subclavian artery and continues to the diaphragm.
- 4. Below the diaphragm and before the bifurcation of the aorta (bifurcation) is the abdominal aorta.
Dividing the aorta into sections is very important for assessing risk and choosing optimal treatment tactics in patients with aortic aneurysms.
An aortic aneurysm is an area of local expansion.
Causes of aortic enlargement
Congenital systemic connective tissue diseases: Marfan syndrome, Ehlers-Danlos syndrome, caused by genetic changes in which the aortic wall has an abnormal structure, can cause the development of an aneurysm.
Acquired diseases that cause aneurysmal changes in the aortic wall: most often this is atherosclerosis. About 80% of all complicated aortic aneurysms are aneurysms caused by an atherosclerotic process, which leads to weakening of the vessel wall and the inability to withstand normal blood pressure, and as a result, to its expansion.
Less commonly, an aortic aneurysm develops in inflammatory diseases caused by external agents (syphilis, fungal infection, tuberculosis) or in autoimmune diseases (nonspecific aortoarteritis).
Major diseases of the aortic arch
Features of the structure and functions of the aortic arch, high pressure and turbulent blood flow contribute to the frequent formation of disorders. The most common pathologies and characteristic changes are presented in the table:
| Disease | Short description |
| Nonspecific aortoarteritis (Takayasu syndrome) | Vasculitis is an inflammatory disease of autoimmune origin. Leads to damage to the vessel, proliferation in the connective tissue and blockage of the lumen |
| "Cervical arch" | Congenital elongation of the aortic arch |
| Atherosclerosis | The appearance of lipid plaques on the vessel wall, prone to destabilization and rupture. Main reasons:
Sclerosis leads to aortic stenosis, dissection and aortic aneurysm |
| Coarctation | A congenital defect manifested in the form of segmental narrowing of the aorta. Most often located in the area where the arc transitions to the descending part. Requires surgical treatment |
| Hypoplasia | Underdevelopment of vascular tissue in the womb. Surgery required |
| Aneurysm | Local dilatation of a section of a vessel due to wall weakness. Requires elective surgical treatment due to risk of sudden rupture and massive internal bleeding |
| Right arc | Violation of organ formation in the embryonic period: the aortic arch turns not to the left, but to the right, and spreads over the right bronchus. In most cases no treatment is required |
| Calcinosis | Accumulation of calcium salts and hardening of the artery wall. The vessel becomes less elastic and fragile, which often leads to ruptures |
| Double arc | Congenital defect characterized by bifurcation of the aorta:
The departments are interconnected. Pathology requires surgical intervention |
| Delamination | Rupture of the aorta at the site of the aneurysm. The prognosis depends on the degree of damage. High mortality rate |
Symptoms of aortic aneurysm
Unfortunately, the diagnosis of aortic aneurysm cannot always be established during the “cold period” (before complications develop), since this disease is usually asymptomatic. Most often, it is discovered accidentally during fluorography, ultrasound or tomography studies performed in connection with other diseases. Treatment of an aneurysm of the ascending aorta before complications develop is much safer for the patient, therefore, in the early diagnosis of an aortic aneurysm, routine medical examination is important.
It is worth noting that every hundredth patient who died suddenly dies from aortic dissection.
Complaints usually appear when the aneurysm begins to stratify or, enlarging, compresses surrounding organs and tissues. Pain or dysfunction of those organs located in the area of the aneurysm appears. At first, this is not of a bright nature and, therefore, does not alarm either the patient or the doctor.
However, the pain intensifies as these deadly complications of an aortic aneurysm develop—it is some of the most severe pain a person can experience. It is localized in the chest if the aneurysm is located in the ascending, descending sections or in its arch, or in the abdomen if it formed in the abdominal section. Characterized by severe weakness, pallor, and often the person loses consciousness.
Impaired blood supply to organs located in the area of aneurysm rupture or aortic dissection (brain or spinal cord, kidneys, intestines, upper or lower extremities) leads to loss of function of these organs, and a large volume of blood loss during aortic rupture represents the most serious danger. To save a life, minutes count. If early surgical treatment is not available, the first-day mortality rate for aortic dissection is 1% per hour (one person in a hundred dies every hour). Within the first 24 hours, 33% of patients die from aortic dissection, 50% of patients within 48 hours and 75% within two weeks. Only early surgical intervention makes it possible to save a significant proportion of patients.
Aorta and its branches
The aorta is the largest arterial vessel in the human body. The aorta has three sections: the ascending part, the arch
and
the descending part.
In the descending part,
the thoracic part (
pars thoracica) and the abdominal
pars abdominalis) of the aorta are distinguished.
The ascending part of the aorta (pars ascendens aortae), about 6 cm long, has an extension in the initial section in the form of a bulb (bulbus aortae), covered with pericardium. Behind the sternum, it goes up and to the right and at the level of the cartilage of the second rib passes into the aortic arch. The right and left coronary arteries depart from the ascending part (in the area of the bulb).
The aortic arch (arcus aortae), convex upward, bends back and to the left and at the level of the III-IV thoracic vertebra passes into the descending part of the aorta. Three large vessels arise from the convex surface of the aortic arch: the brachiocephalic trunk
(truncus brachiocephalicus),
left common carotid artery
(a. carotis communis sinistra) and
left subclavian artery
(a. subclavia sinistra).
The descending part of the aorta (pars descendens aortae; see Fig. 181) is the longest section of the aorta, runs from the level of the IV thoracic vertebra to the IV lumbar vertebra, where it is divided into the right and left common iliac arteries (aortic bifurcation). The descending aorta is divided into thoracic and abdominal parts.
The aorta is located to the left of the midline of the body and with its branches supplies blood to all organs and tissues of the body. The part of it, about 6 cm long, directly leaving the heart and rising upward is called the ascending aorta. It begins with an extension - the bulb - of the aorta, inside which there are three aortic sinuses, located between the inner surface of the aortic wall and the valve flaps. The right and left coronary arteries depart from the aortic bulb. Curving to the left, the aortic arch lies above the pulmonary arteries diverging here, spreads over the beginning of the left main bronchus and passes into the descending part of the aorta. From the concave side of the aortic arch, branches begin to the trachea, bronchi and the thymus; three large vessels depart from the convex side of the arch: on the right is the brachiocephalic trunk, on the left are the left common carotid and left subclavian arteries.
The brachiocephalic trunk, about 3 cm long, extends from the aortic arch and goes up, back and to the right, in front of the trachea. At the level of the right sternoclavicular joint, it is divided into the right common carotid and subclavian arteries. The left common carotid and left subclavian arteries arise directly from the aortic arch to the left of the brachiocephalic trunk.
The common carotid artery (right and left) goes up next to the trachea and esophagus. At the level of the upper edge of the thyroid cartilage, it divides into the external carotid artery, which branches outside the cranial cavity, and the internal carotid artery, which passes inside the skull and goes to the brain.
The external carotid artery is directed upward, passes through the tissue of the parotid gland and in its thickness behind the neck of the condylar process of the mandible is divided into its final branches: the maxillary and superficial temporal arteries. Along its path, the artery gives off lateral branches and supplies blood to the outer parts of the head and neck, mouth and nose, thyroid gland, larynx, tongue, palate, tonsils, sternocleidomastoid and occipital muscles, submandibular, sublingual and parotid salivary glands , skin, bones and muscles of the head (facial and chewing), teeth of the upper and lower jaws, dura mater, outer and middle ear.
The internal carotid artery goes up to the base of the skull without giving off branches, enters the cranial cavity through the canal of the carotid artery in the temporal bone, rises along the carotid groove of the sphenoid bone, lies in the cavernous sinus and, after passing through the dura and arachnoid membranes, is divided into a number of terminal branches . The artery supplies blood to the brain and organ of vision.
The subclavian artery on the left departs directly from the aortic arch, on the right - from the brachiocephalic trunk, goes around the dome of the pleura, passes between the clavicle and the 1st rib, lies in the groove of the same name in the 1st rib, heading towards the armpit. The subclavian artery and its branches supply the cervical spinal cord with
membranes, brain stem, occipital and partly temporal lobes of the cerebral hemispheres, deep and partly superficial muscles of the neck, cervical vertebrae, intercostal muscles of the first and second spaces, part of the muscles of the back of the head, back and shoulder blades, diaphragm, skin of the chest and upper abdomen, rectus abdominal muscle, mammary gland, larynx, trachea, esophagus, thyroid and thymus glands.
At the base of the brain, due to the connection of the anterior cerebral arteries with the anterior communicating artery, as well as the posterior communicating and posterior cerebral arteries, a circular arterial anastomosis is formed - the arterial (Willisian) circle of the cerebrum. The subclavian artery in the axillary region passes into the axillary artery, which lies in the axillary fossa medial to the shoulder joint and humerus next to the vein of the same name, and is surrounded by the trunks of the brachial plexus. The artery supplies blood to the muscles of the shoulder girdle, the skin and muscles of the lateral chest wall, the shoulder and acromioclavicular joints, and the contents of the axillary fossa.
The brachial artery is a continuation of the axillary artery, it passes in the medial groove of the biceps brachii muscle and in the cubital fossa divides into the radial and ulnar arteries. The brachial artery supplies the skin and muscles of the shoulder, humerus and elbow joint.
The radial artery is located laterally on the forearm in the radial groove, parallel to the radius. In the lower section, near its styloid process, the artery is easily palpable, being covered only by skin and fascia. The radial artery passes to the hand under the tendons of the long muscles of the thumb, bends around the back of the first metacarpal bone. It supplies the skin and muscles of the forearm and hand, the radius, elbow and wrist joints.
The ulnar artery is located on the forearm medially in the ulnar groove parallel to the ulna, passing to the palmar surface of the hand. It supplies blood to the skin and muscles of the forearm and hand, ulna, elbow and wrist joints. The ulnar and radial arteries form two arterial networks of the wrist on the hand: dorsal and palmar, feeding ligaments and joints of the wrist, second, third, fourth interosseous spaces and fingers, and two arterial palmar arches - deep and superficial. The superficial palmar arch is formed mainly by the ulnar artery and the superficial palmar branch of the radial artery. Four common palmar digital arteries extend down from the superficial arch, going to the P-III-IV-V fingers. Each of the I, II, III arteries supplies blood to the sides of the II-V fingers facing each other, IV - supplies the ulnar side of the V finger.
The deep palmar arch is located somewhat proximal to the superficial one. It lies under the flexor tendons at the level of the bases of the metacarpal bones. In the formation of the deep palmar arch, the main role belongs to the radial artery, which connects with the deep palmar branch of the ulnar artery. Three palmar metacarpal arteries depart from the deep arch, which go to the second, third and fourth interosseous spaces. These arteries connect with the common palmar digital arteries. Due to the presence of arches and networks anastomosing each other, the blood supply does not suffer during numerous and complex movements of the hand and fingers.
The descending aorta is divided into two parts: thoracic and abdominal.
The thoracic part of the aorta is located asymmetrically on the spine, to the left of the midline and supplies blood to the internal organs located in the chest cavity and its walls. From the thoracic aorta, 10 pairs of posterior intercostal arteries, superior phrenic and splanchnic branches (bronchial, esophageal, pericardial, mediastinal) depart. From the thoracic cavity, the aorta passes into the abdominal cavity through the aortic opening of the diaphragm. Downwards, the aorta gradually shifts medially, especially in the abdominal cavity, and at the site of its division into two common iliac arteries at the level of the IV lumbar vertebra (aortic bifurcation) it is located in the midline and continues in the form of a thin median sacral artery, which corresponds to the caudal artery of mammals. The abdominal part of the aorta supplies blood to the abdominal viscera and abdominal walls.
Visceral and parietal branches depart from the thoracic aorta
Both paired and unpaired vessels depart from the abdominal aorta Among them there are internal and wall ones. The first include three very large unpaired arteries: the celiac trunk, the superior and inferior mesenteric arteries. Paired branches are represented by the middle adrenal, renal and testicular (in women, ovarian arteries). Parietal branches: inferior phrenic, lumbar and inferior median sacral artery.
The celiac trunk departs immediately under the diaphragm at the level of the thoracic vertebra and immediately divides into three branches that supply blood to the abdominal part of the esophagus, stomach, duodenum, pancreas, liver with gall bladder, spleen, lesser and greater omentum.
The superior mesenteric artery arises directly from the abdominal aorta and goes to the root of the mesentery of the small intestine. It gives off a large number of branches that supply blood to the pancreas, small intestine, and right side of the colon, including the right side of the transverse colon.
The inferior mesenteric artery starts from the left semicircle of the abdominal aorta, goes retroperitoneally down and to the left and gives off a number of branches that supply blood to the left part of the transverse colon, descending, sigmoid colon, upper and middle sections of the rectum. The branches of the superior mesenteric artery anastomose with the branches of the celiac trunk and the inferior mesenteric artery, due to which all three large vessels of the abdominal cavity are connected to each other.
The common iliac artery is the largest human artery (with the exception of the aorta). After traveling a certain distance at an acute angle to each other, each of them divides into two arteries: the internal iliac and external iliac.
The internal iliac artery begins from the common iliac artery at the level of the sacroiliac joint, is located retroperitoneally, goes to the small pelvis, adjacent to its lateral wall. The internal iliac artery supplies the pelvic bone, sacrum and the entire mass of muscles of the small, large pelvis, gluteal region and partly the adductor muscles of the thigh, as well as the viscera located in the small pelvis: rectum, bladder; in men - seminal vesicles, vas deferens, prostate gland; in women - the uterus and vagina, external genitalia and perineum.
The external iliac artery begins at the level of the sacroiliac joint from the common iliac artery, goes retroperitoneally down and forward, passes under the inguinal ligament and passes into the femoral artery. The external iliac artery supplies blood to the thigh muscles, the scrotum in men, and the pubis and labia majora in women.
The femoral artery is a direct continuation of the external iliac artery. It passes in the femoral triangle, between the thigh muscles, enters the popliteal fossa, where it continues into the popliteal artery. The femoral artery supplies blood to the femur, skin and muscles of the thigh, skin of the anterior abdominal wall, external genitalia, and hip joint.
The popliteal artery is a continuation of the femoral artery. It lies in the fossa of the same name, passes to the lower leg, where it immediately divides into the anterior and posterior tibial arteries. The artery supplies blood to the skin and nearby muscles of the thigh and back of the leg, and the knee joint.
The posterior tibial artery goes down, in the area of the ankle joint it passes to the sole behind the medial malleolus under the flexor retinaculum, after which it divides into its terminal branches: the medial and lateral plantar arteries. The largest branch of the posterior tibial is the peroneal artery. The posterior tibial artery supplies the skin of the back of the leg, bones, leg muscles, knee and ankle joints, and foot muscles.
The anterior tibial artery descends down the anterior surface of the interosseous membrane of the leg. The artery supplies the skin and muscles of the anterior surface of the leg and the back of the foot, the knee and ankle joints, and on the foot it passes into the dorsal artery of the foot. Both tibial arteries form a plantar arterial arch on the foot, which lies at the level of the bases of the metatarsal bones. Arteries that supply the skin and muscles of the foot and toes extend from the arch.
Diagnosis of aortic aneurysm
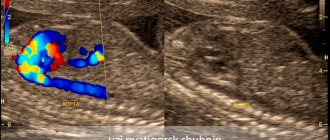
In the diagnosis of aortic aneurysms, the so-called imaging techniques (ultrasound, MRI, CT, AG) are of greatest importance. In the ascending aorta, its arch and in the abdominal section, an aneurysm can be detected using ultrasound methods (ultrasound). To diagnose an aneurysm of the descending (thoracic) aorta, X-ray methods (x-ray, computed tomography) are required. To establish a final diagnosis and select a treatment method, contrast research methods are performed. Currently, the optimal diagnostic method, which provides the most complete information about the location, extent, diameter of the aneurysm and its relationship to nearby organs, is multislice computed tomography - aortography.
The structure of the aorta and its branches
The aorta is the largest unpaired arterial vessel in the systemic circulation. The aorta is divided into three sections: the ascending aorta, the aortic arch and the descending aorta, which in turn is divided into the thoracic and abdominal parts.
The ascending aorta exits the left ventricle behind the left sternal border at the level of the third intercostal space; in the initial section it has an extension - the aortic bulb (25-30 mm in diameter).
At the location of the aortic valve, there are three sinuses on the inside of the aorta. Each of them is located between the corresponding semilunar valve and the aortic wall. The right and left coronary arteries depart from the beginning of the ascending aorta. The ascending part of the aorta lies behind and partly to the right of the pulmonary trunk, rises upward and, at the level of the junction of the 2nd right costal cartilage with the sternum, passes into the aortic arch (here its diameter decreases to 21-22 mm).
The aortic arch turns left and back from the posterior surface of the 2nd costal cartilage to the left side of the body of the 4th thoracic vertebra, where it passes into the descending part of the aorta.
In this place there is a slight narrowing - an isthmus. The edges of the corresponding pleural sacs approach the anterior semicircle of the aorta on its right and left sides. The left brachiocephalic vein is adjacent to the convex side of the aortic arch and to the initial sections of the large vessels extending from it (brachiocephalic trunk, left common carotid and subclavian arteries), and the right pulmonary artery begins under the aortic arch, below and slightly to the left is the bifurcation of the pulmonary trunk.
Behind the aortic arch is the bifurcation of the trachea. Between the bent semicircle of the aortic arch and the pulmonary trunk or the beginning of the left pulmonary artery there is an arterial ligament. At this point, thin arteries extend from the aortic arch to the trachea and bronchi.
Three large arteries begin from the convex semicircle of the aortic arch: the brachiocephalic trunk, the left common carotid and the left subclavian arteries.
The descending aorta is the longest section of the aorta, running from the level of the 4th thoracic vertebra to the 4th lumbar vertebra, where it divides into the right and left common iliac arteries; this location is called the aortic bifurcation.
The descending aorta is in turn divided into thoracic and abdominal parts.
The thoracic aorta is located in the chest cavity in the posterior mediastinum. Its upper section is located in front and to the left of the esophagus. Then, at the level of 8-9 thoracic vertebrae, the aorta bends around the esophagus on the left and goes to its posterior surface. To the right of the thoracic part of the aorta are the azygos vein and the thoracic duct, to the left is the parietal pleura adjacent to it, at the point where it passes into the posterior part of the left mediastinal pleura.
In the chest cavity, the thoracic part of the aorta gives off paired parietal branches; posterior intercostal arteries, as well as visceral branches to the organs of the posterior mediastinum.
The abdominal part of the aorta , being a continuation of the thoracic part of the aorta, begins at the level of the 12th thoracic vertebra, passes through the aortic opening of the diaphragm and continues to the level of the middle of the body of the 4th lumbar vertebra. The abdominal part of the aorta is located on the anterior surface of the lumbar vertebral bodies, to the left of the midline; lies retroperitoneally.
To the right of the abdominal aorta are the inferior vena cava, anteriorly the pancreas, the horizontal (lower) part of the duodenum and the root of the mesentery of the small intestine. The abdominal part of the aorta gives off paired parietal branches to the diaphragm and to the walls of the abdominal cavity, and itself directly continues into the thin median sacral artery.
The visceral branches of the abdominal aorta are the celiac trunk, the superior and inferior mesenteric arteries (unpaired branches) and the paired renal, middle adrenal and ovarian arteries.
Right and left coronary arteries (see above);
Treatment methods for aortic aneurysm
The main method of treating an aneurysm of any part of the aorta is surgical. The point of the method is to replace the dilated section of the aorta in order to prevent its further stretching and rupture. To replace the aorta, two methods are used - the endovascular (intravascular) method using a special intravascular prosthesis (stent graft), and open surgery - aortic replacement.
Each method has its own indications, and each of them has its own advantages and disadvantages.
The advantages of the surgical method lie in its versatility, that is, the ability to correct all disorders associated with an aortic aneurysm, regardless of the department and nature of the lesion. For example, in case of an aneurysm of the ascending aorta and damage to the aortic valve, aortic and aortic valve replacement is performed in combination with coronary bypass surgery.
To perform surgery on the ascending aorta and its arch, it is necessary to use artificial circulation, systemic hypothermia, and often complete circulatory arrest.
Indications for surgical treatment
- transverse size of the aneurysm,
- aneurysm growth rate;
- the formation of complications of this disease.
For each section of the aorta, there is a cutoff limit for the transverse size of the aorta, after which the risk of aortic rupture statistically significantly increases. Thus, for the ascending and abdominal aorta, the transverse diameter of the aneurysm is 5 cm dangerous in terms of rupture, for the thoracic aorta - 6 cm. If the diameter of the aneurysm increases by more than 6 mm in 6 months, then this is also an indication for surgery. Also threatening in terms of rupture and dissection of the aorta are the saccular form of the aneurysm and expansion of the aorta, which is smaller than the diameter that is the indication for surgery, but accompanied by pain at the site of expansion and dysfunction of the presenting organs. Dissections and completed ruptures of aneurysms are absolute indications for emergency surgery.
Acute traumatic aortic injury: imaging and management
Acute traumatic injury of the aorta (ATI) is fatal in most cases - with such damage, about 80% of victims die before arriving at the hospital. Among the causes of death as a result of road accidents, traumatic brain injury is second only to traumatic brain injury in frequency. With improved imaging options and advances in surgical treatments and percutaneous interventions, mortality among patients with OTPA has decreased from 50% to 5%. OTPA occurs in approximately 2% of blunt chest trauma cases, approximately 70% of which occur in motor vehicle accident victims.
The exact pathogenesis of OTPA is not entirely clear, but the proposed mechanisms are a sharp increase in intravascular pressure (like a water hammer effect) and rupture of the aorta, sandwiched between the vertebral body and the sternum displaced towards the spine (Fig. 1). .
Figure 1 | Mechanisms of aortic injury in patients with blunt chest trauma
The method of choice for diagnosing OTPA is CT angiography, which replaces conventional angiography. It allows you to evaluate intimal disorders and indirect signs of aortic damage, such as periaortic hematoma. In emergency settings, performing ECG-gated CT to visualize the aorta is impractical due to lengthy setup times, the need for additional preparation, and prolonged patient breath holding. With the advent of new dual-source CT scanners with ultra-fast acquisition speeds, it has become possible to obtain virtually “still” images without the motion artifacts seen in the heart and aortic root with traditional CT scanning. With these images, radiological assessment of OTPA can be used to determine patient management and prognosis.
Next, we will describe current aspects of imaging, radiological classification, diagnosis and management of patients with OTPA, as well as recent advances that have a significant impact on the accuracy of imaging in patients with this injury.
Due to its diagnostic capabilities and availability of the method, CT angiography is the study of choice for diagnosing OTPA. The most commonly injured areas are the aortic isthmus, ascending aorta, and aortic hiatus, and arterial phase CT scans of the chest and upper abdomen should be obtained in patients with blunt trauma. Most often, CT angiography is performed as part of a general computed tomography when examining patients with polytrauma.
The advent of dual-source CT scanners and scanners with 64 or more slices has revolutionized the use of CT in cardiac and vascular imaging. These scanners have spatial resolutions ranging from 0.25 to 0.47 mm, with most new scanners providing a resolution of 0.33 mm. Also, such scanners have very fast gantry rotation, which leads to a higher speed of image acquisition - still images of even the ascending aorta can be obtained without ECG synchronization (Fig. 2). In addition, motion artifacts from the patient's breathing are minimal, which provides the necessary diagnostic image quality.
Figure 2
ECG-unsynchronized axial CT image at the level of the sinuses of Valsalva (the space between the semilunar valves and the aortic wall) obtained using a new dual-source CT scanner (A) MSCT on a 64-slice scanner in the same patient. Note the absence of ripple artifacts (indicated by arrows) in the new dual-source scanner, even without ECG synchronization.
The rationale for arterial phase imaging of the chest and upper abdomen is that most vascular injuries anatomically occur in these areas unless the patient has additional risk factors such as a pelvic fracture and therefore possible pelvic vascular injury. In addition, arterial phase imaging of the chest alone does not preclude the detection of any serious pulmonary pathologies.
A low-dose delayed phase scan of the abdomen and pelvis is performed if damage is confirmed during the portal venous phase of the study or if kidney or bladder damage is suspected. An initial noncontrast phase is useful in diagnosing intramural hematoma, but is not performed as part of the trauma protocol. The chest and abdominal CT protocol for patients with blunt trauma is shown in Table 1.
Table 1 | CT examination protocol for blunt trauma of the chest, abdomen and pelvis
In each case, sagittal and coronal 3D models are generated through images in the axial plane. Other 3D methods, including maximum intensity projection (MIP) and volume rendering (VR), are created on a separate workstation. Curved aortic reconstructions are created only in cases where stent placement is planned later during surgery. This 3D data modeling is always read in combination with raw data, which greatly assists in surgical planning.
MRI is almost never used to identify injuries in patients with blunt trauma. However, MR angiography is also used to diagnose trauma: this method is mainly used after placement of an endovascular stent or if renal failure precludes CT angiography. The MRI protocol for aortic imaging is highlighted in Table 2. If CT and MR angiography cannot be performed due to renal failure (eGFR < 30), patients are best treated with non-contrast CT for interval changes in aortic diameter.
Table 2 | Protocol of MR angiography of the aorta
Although CT angiography is the definitive modality in most cases, the initial evaluation of injury often begins with a chest x-ray. Data suspicious for OTPA with this research method include:
- mediastinal widening of 8 cm at the level of the aortic arch (or superior mediastinum, measuring approximately 25% of the width of the chest);
- uneven contour of the aortic arch;
- inability to determine the aortopulmonary window;
- darkening of the border of the lung with the aorta;
- deviation of the trachea or nasogastric tube to the right;
- omission of the left main bronchus;
- dilated left paratracheal stripe.
CT angiography has very high sensitivity (approximately 98%) and almost 100% specificity for diagnosis. Signs of OTPA on CT angiography can be divided into direct and indirect.
Indirect signs of PAAD include mediastinal or periaortic hematoma, retrocrural hematoma, or a small caliber of the aorta distal to the injury site. The location of the mediastinal hematoma is critical because if there is a clear layer of fatty tissue between the aorta and the hematoma, alternative sources of bleeding (from a vein, intercostal artery, or bone) should be considered (Figure 3). Periaortic hematoma without any obvious signs of aortic injury may be associated with occult intimal injury. Conventional angiography may not provide sufficient diagnostic imaging in patients with occult intimal injury, so additional use of intravascular ultrasound or transesophageal echocardiography is better. .
Figure 3
A 36-year-old man presented with a mediastinal hematoma and hemothorax. Sagittal (A) and coronal images (B, C) of CT angiography. A clearly defined layer of adipose tissue is identified between the descending aorta and the hematoma (indicated by arrows). This suggests that the aorta is not the source of the hematoma. The source of ongoing bleeding is the intercostal artery (arrow in image C)
Direct signs of OTPA include active contrast extravasation, rupture (or traumatic pseudoaneurysm), intramural thrombus, aortic displacement, and aortic contour abnormality (including sudden change in caliber, also known as pseudocoarctation). When there is a high risk of a mechanism of injury, as in a motor vehicle injury, evaluation of other associated conditions is also critical. Typically, PAH is accompanied by severe head injuries, damage to the lungs and heart, rupture of the diaphragm, intra-abdominal bleeding, and fractures of the pelvis and long bones.
The term minimal aortic injury (MAI) is an evolving concept that was introduced in 1999 by the Presley Center Aortic Trauma CT Grading System (Table 3) to describe injuries that are minor in nature.
Malhotra et al. were the first to describe this injury and defined it as a small (<1 cm) post-traumatic intimal valve with minimal or no periaortic hematoma. Most of these injuries cannot be diagnosed by other imaging modalities before routine use of CT angiography. MPA was initially found in approximately 10% of patients with OTPA, but more recent studies have shown that it occurs in approximately 25% to 35% of OTPA cases. The increase in the detection rate of MPA may be due to a number of factors, such as increased availability of CT angiography, improved CT technology, and improved vehicle safety. The descending aorta is the most common location of MPA, with the aortic isthmus being the second most common site. Although the generally accepted criterion for MPA is minimal or absent mediastinal hematoma, there is no standard finding that defines MPA. Variations in the definition of minimal or negligible aortic injury in various studies are given in Table 3.
Table 3 | Definition of the group of patients with minor/minimal aortic injuries in various studies
The use of different signs of minimal or minor aortic injury is misleading to radiologists and surgeons, as this may influence tactical decisions. Among the key features, most studies considered a “small” intimate flap, intraluminal thrombus, or intramural hematoma without any external contour abnormalities and with minimal or no periaortic hematoma as MPA (Figs. 4, 5). In most studies, an intimal flap was considered “small” if it measured less than 1 cm, as untreated intimal flaps or thrombi larger than 1 cm had a higher incidence of disease progression. .
Figure 4
Frontal (A) and sagittal (B) CT images of the chest of a 32-year-old man (jumped from the roof of a building). A tiny intraluminal thrombus (arrows) is identified in the aorta at the level of the isthmus, without external hematoma or irregular contour shape suggesting aortic damage.
Figure 5
Axial CT image of the chest in a 41-year-old man (fall from a motorcycle). A small intimal flap (indicated by a black arrow) is identified in the anterior descending thoracic aorta without external hematoma or deformation of the external contour, which corresponds to minimal damage to the aorta. We also see a fracture of the left rib (indicated by a white arrow) and bilateral accumulation of fluid in the pleural cavity.
As with the diagnosis of MPA, there is also no consensus regarding the management and monitoring of these injuries. Most studies suggest that they can be treated noninvasively by monitoring heart rate and blood pressure and anticoagulant or antiplatelet therapy. The choice of appropriate treatment strategy should be individualized, taking into account other concomitant diseases. The initial use of negative inotropes (most often beta-blockers) is suggested to achieve target blood pressure and heart rate values to reduce vascular shear stress—systolic blood pressure of 120 mmHg. Art. or an average blood pressure of 80 mm Hg. Art., as well as a heart rate less than 80 beats per minute. Second-line drugs are calcium channel blockers or arterial vasodilators. The Society of Vascular Surgery (SVS) guidelines suggest a “watchful waiting” approach with serial monitoring of the intimal condition.
Recent Eastern Association for the Surgery of Trauma (EAST) guidelines note that there is insufficient evidence to make recommendations for the conservative treatment of MPA, with more research needed. Even the SVS guidelines lack any specific recommendations on the “expected” course of the disease and follow-up protocol. Visual observation of MPA usually varies depending on the experience of each individual institution and its ongoing observations. Although a recent study from Harborview Medical Center by Heneghan et al. suggest that after conservative treatment of MPA there is no need for follow-up imaging, a previous study from the same institute in 2014, Gunn et al. suggested the use of CT angiography on day 3, 1 week, and 1 month after injury. In another study, Mosquera et al. recommended CT angiography at 1, 3, 6, and 12 months after injury, followed by annual cardiac and vascular MR scanning. Although there is no consensus regarding the timing of prophylactic testing, most investigators suggest that surveillance usually ends when the aorta returns to normal appearance. Almost all studies show that in most patients the disease does not progress, and if it does, it occurs within the first month after injury. This pattern of disease progression is also supported by experimental animal studies, which show that maximum intimal cellular activity and thickening occur 3–4 weeks after injury. If after this time there are signs of stabilization or improvement in the imaging appearance of the injury, further observation may not be necessary. Ultimately, there is a need for a prospective multicenter study using a “fixed” anatomical definition of “minor” aortic injury, in which a cohort of patients with MPA should be serially monitored with a predetermined CT frequency and duration of follow-up.
There are several different OTPA classification systems (Table 4). One of the most widely used OTPA assessments was proposed by Azizzadeh et al. (approved by the Society of Vascular Surgery) and divides injuries into 4 degrees: class 1 - intimal tears (Fig. 5), class 2 - intrathoracic hematoma or large intimal valve (Fig. 6), class 3 - pseudoaneurysm (Fig. 7, and 4 grade—free tear (Figure 9).Although this grading system provides a straightforward description of the injury, it is not a guide to treatment.
One of the most widely used OTPA assessments was proposed by Azizzadeh et al. (approved by the Society of Vascular Surgery) and divides injuries into 4 degrees: class 1 - intimal tears (Fig. 5), class 2 - intrathoracic hematoma or large intimal valve (Fig. 6), class 3 - pseudoaneurysm (Fig. 7, and 4 grade—free tear (Figure 9).Although this grading system provides a straightforward description of the injury, it is not a guide to treatment.
Table 4 | OTPA classifications
Figure 6
A 45-year-old man with blunt chest trauma. Axial (A), coronal (B) and sagittal (C) planes of CT angiography image. A PA is detected in the proximal descending aorta, causing an intramural hematoma in the area of the aortic arch, branches of the aortic arch (curved arrow in image C) and the descending thoracic aorta (white arrows) with thrombus (hollow arrows in images A and C).
Figure 7
A 63-year-old woman suffered as a result of a motorcycle injury. Axial (A) and sagittal (B) sections of CT scan of the chest. A small pseudoaneurysm (indicated by white arrows) is identified in the area of the aortic isthmus with a mediastinal hematoma (indicated by a hollow arrow).
Figure 8
Male, 33 years old, OTPA. Axial (A) and coronal (B) images of CT angiography with ECG synchronization. A pseudoaneurysm of the ascending thoracic aorta is visualized from the area above the sinotubular junction. The dimensions of the pseudoaneurysm are 1.4 x 0.7 cm. There are no signs of active contrast extravasation. An open arrow indicates slight bleeding from mediastinal fat.
Figure 9
A 70-year-old woman with a traumatic aortic rupture. Axial (A) and coronal (B) CT images. There are signs of complete rupture of the anterior wall of the descending thoracic aorta (indicated by an arrow), causing extravasation of contrast material (indicated by an asterisk). Note the mediastinal hematoma and bilateral fluid collections in the pleural cavities.
There are many treatment options for a patient with OTPA, such as traditional open surgery, endovascular stenting, and the choice of conservative management for some groups of patients. Thus, evaluation of CT angiography of the OTPA is highly valuable for treatment planning. In addition to information about the severity and type of injury, CT findings should include a description of the aortic arch anatomy, signs of vertebral artery dominance, any significant pre-existing atherosclerotic disease or stenosis, previous postoperative changes (such as coronary artery bypass graft), length of aortic injury, greater aortic diameter and below the injury, as well as the proximity of the left subclavian artery (in case of injury to the aortic isthmus). This information helps the doctor choose treatment options and type of surgery. For example, surgical repair of the aorta typically involves a left posterolateral thoracotomy, often requiring the patient to be placed on a heart-lung machine. Imaging helps the surgeon determine the exact area of injury and provides information about other injuries that can be treated using the same approach.
A few additional important radiological findings to consider when planning endovascular stenting are the presence of any focal lesion or thrombus, information about the proximal and distal stent site (preferred length is 2 cm), the presence of calcification in the stent site, aortic tortuosity and diameter of access vessels.
There are differing opinions regarding the best management option for patients with this type of injury. Studies have shown that the increased use of endovascular stents among patients requiring surgical treatment has led to a decrease in the incidence of early complications and shorter hospital stays. Recent EAST guidelines recommend the use of endovascular grafting in patients who have no contraindications to the procedure.
The guidelines also suggest that in hemodynamically stable patients, definitive treatment with OTPA should be delayed until other acute life-threatening injuries have resolved. Early treatment with OTPA may be associated with a higher incidence of paraplegia and renal failure. But if the patient is hemodynamically unstable and there is direct evidence of aortic injury on CT, urgent treatment of the aortic injury is necessary. The proposed management algorithm for OTPA in patients with blunt trauma, based on the recommendations of the EAST Practice Work Group, is shown in Table 5.
Table 5 | Tactics for OTPA, based on the recommendations of the Eastern Association of Surgeons and Traumatologists (EAST practice work group)
Analysis of a national inpatient sample by Ultee et al. showed that treatment using endovascular methods (TEVAR - thoracic endovascular aortic repair) increased from 6.5% of surgical volume in 2005 to 87% in 2011. Overall mortality from aortic trauma has also decreased, due to the use of TEVAR, expanded indications for use, improved surgeon skills, and increased technical capabilities. Aortic injury, relatively common in younger age groups, presents many unique challenges related to its management and outcome. The average aortic size in this population is much smaller than in aneurysmal disease, and therefore early experience with endovascular aortic repair (EVAR) encountered problems in determining graft size. The arch geometry in younger patients is also more acute, which is associated with poor compatibility of the endograft with the aortic arch. A 0–18% rate of infolding, migration, stent collapse, and even acute aortic occlusion requiring reintervention has been reported, with most complications occurring early after surgery. Aortic remodeling in these patients may also differ in that the aorta distal to the left subclavian artery dilates at a faster rate than the proximal segments. In addition to technical advances, the use of procedural modifications, such as limiting excess graft size to 10% to accommodate subsequent remodeling, has been proposed. The influence of these factors on long-term results requires further study.
With hybrid operating rooms becoming more common, parallel advances in imaging such as cone beam CT, fusion imaging, and intravessel ultrasound (IVUS) are also facilitating operative and interventional procedures by minimizing radiation exposure and use of contrast agents. This, along with better image quality, is critical to the technical success of complex endovascular aortic procedures. Better planning software and fusion visualization are helpful for accurate graft deployment and this can be further confirmed by IVUS. The use of IVUS in aortic procedures improves understanding of complex anatomy and has been shown to reduce the amount of contrast used and overall operative time.
The patient with blunt trauma requires a multidisciplinary approach to treatment. Because OTPA is a life-threatening condition with almost always unpredictable outcomes, multidisciplinary teams are needed to rapidly assess, triage, and manage patients suspected of having OTPA (based on physical examination, external or CT findings, or known prior history). This group includes specialists in cardiovascular imaging, interventional radiology, cardiac surgery, vascular and endovascular surgery, cardiology, and anesthesiology and resuscitation. The team should provide expert judgment for all acute situations and discuss various options for approaches to the treatment of OTPA, including the use of endovascular techniques, minimally invasive and open surgery. In the researchers' experience, it is the interdisciplinary team approach to the management of patients with OTPA that is critical for appropriate treatment.
Sources:
- Fox N, Schwartz D, Salazar JH, et al. Evaluation and management of blunt traumatic aortic injury: a practice management guideline from the Eastern Association for the Surgery of Trauma. J Trauma Acute Care Surg. 2015;78(1):136–46.
- Neschis DG, Scalea TM, Flinn WR, Griffith BP. Blunt aortic injury. N Engl J Med. 2008;359(16):1708–16.
- Heneghan RE, Aarabi S, Quiroga E, Gunn ML, Singh N, Starnes BW. Call for a new classification system and treatment strategy in blunt aortic injury. J Vasc Surg. 2016;64(1):171–6.
- Ungar TC, Wolf SJ, Haukoos JS, Dyer DS, Moore EE. Derivation of a clinical decision rule to exclude thoracic aortic imaging in patients with blunt chest trauma after motor vehicle collisions. J Trauma. 2006;61(5):1150–5.
- Steenburg SD, Ravenel JG, Ikonomidis JS, Schonholz C, Reeves S. Acute traumatic aortic injury: imaging evaluation and management. Radiology. 2008;248(3):748–62.
- Nagpal P, Khandelwal A, Saboo SS, Bathla G, Steigner ML, Rybicki FJ. Modern imaging techniques: applications in the management of acute aortic pathologies. Postgrad Med J 1078;2015(91):449–62.
- Seimens Healthineers USA
- Starnes BW, Lundgren RS, Gunn M, et al. A new classification scheme for treating blunt aortic injury. J Vasc Surg. 2012;55(1): 47–54.123
- Lamarche Y, Berger FH, Nicolaou S, et al. Vancouver simplified grading system with computed tomographic angiography for blunt aortic injury. J Thorac Cardiovasc Surg 2012;144(2):347–54.
- Rogalla P, Kloeters C, Hein PA. CT technology overview: 64-slice and beyond. Radiol Clin North Am. 2009;47(1):1–11.
- Voitle E, Hofmann W, Cejna M. Aortic emergencies-diagnosis and treatment: a pictorial review. Insights Imaging. 2015;6(1):17–32.
- Nagpal P, Saboo SS, Khandelwal A, Duran-Mendicuti MA, Abbara S, Steigner ML. Traumatic right atrial pseudoaneurysm. Cardiovasc Diagnosis Ther. 2015;5(2):141–4.
- Soto JA, Anderson SW. Multidetector CT of blunt abdominal trauma. Radiology. 2012;265(3):678–93.
- Fishman EK, Ney DR, Heath DG, Corl FM, Horton KM, Johnson PT. Volume rendering versus maximum intensity projection in CT angiography: what works best, when, and why. Radiographics. 2006;26(3):905–22.
- Sammer M, Wang E, Blackmore CC, Burdick TR, Hollingworth W. Indeterminate CT angiography in blunt thoracic trauma: is CT angiography enough? AJR Am J Roentgenol. 2007;189(3):603–8.
- Fabian TC, Richardson JD, Croce MA, et al. Prospective study of blunt aortic injury: Multicenter Trial of the American Association for the Surgery of Trauma. J Trauma.1997;42(3):374–80 (discussion 80–83).
- Kram HB, Appel PL, Wohlmuth DA, Shoemaker WC. Diagnosis of traumatic thoracic aortic rupture: a 10-year retrospective analysis. Ann Thorac Surg. 1989;47(2):282–6.
- Gavant ML. Helical CT grading of traumatic aortic injuries. Impact on clinical guidelines for medical and surgical management. Radiol Clin North Am. 1999;37(3):553–74.
- Malhotra AK, Fabian TC, Croce MA, Weiman DS, Gavant ML, Pate JW. Minimal aortic injury: a lesion associated with advanced diagnostic techniques. J Trauma.2001;51(6):1042–8.
- Forman MJ, Mirvis SE, Hollander DS. Blunt thoracic aortic injuries: CT characterization and treatment outcomes of minor injury. Eur Radiol. 2013;23(11):2988–95.
- Gunn ML, Lehnert BE, Lungren RS, et al. Minimal aortic injury of the thoracic aorta: imaging appearances and outcome. Emerg Radiol. 2014;21(3):227–33.
- Paul JS, Neideen T, Tutton S, et al. Minimal aortic injury after blunt trauma: selective nonoperative management is safe. J Trauma. 2011;71(6):1519–23.
- de Mestral C, Dueck A, Sharma SS, et al. Evolution of the incidence, management, and mortality of blunt thoracic aortic injury: a population-based analysis. J Am Coll Surg. 2013;216(6):1110–5.
- Osgood MJ, Heck JM, Rellinger EJ, et al. Natural history of grade I–II blunt traumatic aortic injury. J Vasc Surg. 2014;59(2):334–41.
- Kidane B, Abramowitz D, Harris JR, DeRose G, Forbes TL. Natural history of minimal aortic injury following blunt thoracic aortic trauma. Can J Surg Journal canadien de chirurgie.2012;55(6):377–81.
- Mosquera VX, Marini M, Lopez-Perez JM, et al. Role of conservative management in traumatic aortic injury: comparison of long-term results of conservative, surgical, and endovascular treatment. J Thorac Cardiovasc Surg. 2011;142(3):614–21.
- Erbel R, Aboyans V, Boileau C, et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J 2014;35(41):2873–926.
- Lee WA, Matsumura JS, Mitchell RS, et al. Endovascular repair of traumatic thoracic aortic injury: clinical practice guidelines of the Society for Vascular Surgery. J Vasc Surg. 2011;53(1):187–92.
- Neville RF, Padberg FT Jr, DeFouw D, Hernandez J, Duran W, Hobson RW 2nd. The arterial wall response to intimal injury in an experimental model. Ann Vasc Surg 1992;6(1):50–4.
- Azizzadeh A, Keyhani K, Miller CC 3rd, Coogan SM, Safi HJ, Estrera AL. Blunt traumatic aortic injury: initial experience with endovascular repair. J Vasc Surg. 2009;49(6):1403–8.
- Forcillo J, Philie M, Ojanguren A, et al. Outcomes of traumatic aortic injury in a primary open surgical approach paradigm. Trauma Mon. 2015;20(2):e18198.
- Fanelli F, Dake MD. Standard of practice for the endovascular treatment of thoracic aortic aneurysms and type B dissections. Cardiovasc Intervent Radiol. 2009;32(5):849–60.
- Ultee KH, Soden PA, Chien V, et al. National trends in utilization and outcome of thoracic endovascular aortic repair for traumatic thoracic aortic injuries. J Vasc Surg.2016;63(5):1232–9.
- Forbes TL, Harris JR, Lawlor DK, Derose G. Aortic dilatation after endovascular repair of blunt traumatic thoracic aortic injuries. J Vasc Surg. 2010;52(1):45–8.
- Farber MA, Mendes RR. Endovascular repair of blunt thoracic aortic injury: techniques and tips. J Vasc Surg. 2009;50(3):683–6. 36. Kolbel T, Dias N, Resch T, Holst J, Sonesson B, Malina M. In situ bending of thoracic stent grafts: clinical application of a novel technique to improve conformance to the aortic arch.
- J Vasc Surg. 2009;49(6):1613–6.
- Neschis DG, Moainie S, Flinn WR, Scalea TM, Bartlett ST, Griffith BP. Endograft repair of traumatic aortic injury—a technology in evolution: a single institution's experience. Ann Surg. 2009;250(3):377–82.
- Stangenberg L, Shuja F, Carelsen B, Elenbaas T, Wyers MC, Schermerhorn ML. A novel tool for three-dimensional roadmap- ping reduces radiation exposure and contrast agent dose in complex endovascular interventions. J Vasc Surg. 2015;62(2):448–55.
- Song TK, Donayre CE, Kopchok GE, White RA. Intravascular ultrasound use in the treatment of thoracoabdominal dissections, aneurysms, and transections. Semin Vasc Surg. 2006;19(3):145–9.
- Guo BL, Shi ZY, Guo DQ, et al. Effect of intravascular ultrasound-assisted thoracic endovascular aortic repair for ''complicated'' type B aortic dissection. Chin Med J (Engl). 2015;128(17):2322–9.
- Dijkstra ML, Eagleton MJ, Greenberg RK, Mastracci T, Hernandez A. Intraoperative C-arm cone-beam computed tomography in fenestrated/branched aortic endografting. J Vasc Surg. 2011;53(3):583–90.
- Bashore TM, Bates ER, Berger PB, et al. American College of Cardiology/Society for Cardiac Angiography and Interventions Clinical Expert Consensus Document on cardiac catheterization laboratory standards. A report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2001;37(8):2170–214.
- Alberta HB, Secor JL, Smits TC, et al. Comparison of thoracic aortic diameter changes after endograft placement in patients with traumatic and aneurysmal disease. J Vasc Surg. 2014;59(5):1241–6.
- Watson J, Slaiby J, Garcia Toca M, Marcaccio EJ Jr, Chong TT. A 14-year experience with blunt thoracic aortic injury. J Vasc Surg. 2013;58(2):380–5.
- Fearn S, Lawrence-Brown MM, Semmens JB, Hartley D. Follow-up after endovascular aortic aneurysm repair: the plain radiograph has an essential role in surveillance. J Endovasc Ther. 2003;10(5):894–901.
- Riesenman PJ, Brooks JD, Farber MA. Acute blunt traumatic injury to the descending thoracic aorta. J Vasc Surg. 2012;56(5):1274–80. 47. Estrera AL, Miller CC 3rd, Guajardo-Salinas G, et al. Update on blunt thoracic aortic injury: fifteen-year single-institution experience. J Thorac Cardiovasc Surg. 2013;145(3 Suppl):S154–8. 48. Rabin J, DuBose J, Sliker CW, O'Connor JV, Scalea TM, Griffith BP. Parameters for successful nonoperative management of traumatic aortic injury. J Thorac Cardiovasc Surg. 2014;147(1):143–9.
Types of open surgical operations for aortic aneurysms:
- Bentalla-De Bono operation (replacement of the ascending aorta with a valve-containing conduit with a mechanical prosthetic aortic valve);
- David's operation (replacement of the ascending aorta while preserving the native aortic valve);
- Supracoronary aortic replacement;
- Prosthetics of the ascending aorta and its arch (Borst technique, the use of oblique aggressive anastomosis and other techniques);
- Thoracic aortic replacement;
- Abdominal aortic replacement.
Aorta and its parts. Branches of the aortic arch, their anatomy, topography, areas of branching (blood supply)
Aorta, aorta is the largest unpaired arterial vessel of the systemic circulation. The aorta is divided into three sections: the ascending aorta, the aortic arch and the descending aorta, which in turn is divided into the thoracic and abdominal parts.
Ascending aorta, pars ascendens aortae,
exits the left ventricle behind the left edge of the sternum at the level of the third intercostal space;
in the initial section it has an extension - the aortic bulb, bulbus aortae
(25-30 mm in diameter).
At the location of the aortic valve, there are three sinuses, sinus aortae, on the inner side of the aorta.
Each of them is located between the corresponding semilunar valve and the aortic wall. The right and left coronary arteries depart from the beginning of the ascending aorta.
The ascending part of the aorta lies behind and partly to the right of the pulmonary trunk, rises upward and, at the level of the junction of the second right costal cartilage with the sternum, passes into the aortic arch (here its diameter decreases to 21-22 mm).
Aortic arch, arcus aortae,
turns to the left and back from the posterior surface of the second costal cartilage to the left side of the body of the fourth thoracic vertebra, where it passes into the descending part of the aorta.
In this place there is a slight narrowing - the isthmus of the aorta, isthmus aortae.
The edges of the corresponding pleural sacs approach the anterior semicircle of the aorta on its right and left sides.
The left brachiocephalic vein is adjacent to the convex side of the aortic arch and to the initial sections of the large vessels extending from it (brachiocephalic trunk, left common carotid and subclavian arteries), and the right pulmonary artery begins under the aortic arch, below and slightly to the left is the bifurcation of the pulmonary trunk. Behind the aortic arch is the bifurcation of the trachea.
Between the concave semicircle of the aortic arch and the pulmonary trunk or the beginning of the left pulmonary artery there is an arterial ligament, tig.
arteriosum. At this point, thin arteries extend from the aortic arch to the trachea and bronchi. Three large arteries begin from the convex semicircle of the aortic arch: the brachiocephalic trunk, the left common carotid and the left subclavian arteries.
Descending part of the aorta, pars descendens aortae,
- this is the longest section of the aorta, passing from the level of the IV thoracic vertebra to the IV lumbar vertebra, where it is divided into the right and left common iliac arteries;
this place is called the bifurcation of the aorta, bifurcdtio aortae.
The descending aorta is in turn divided into thoracic and abdominal parts.
Thoracic part of the aorta, pars thordcica aortae,
located in the chest cavity in the posterior mediastinum.
Its upper section is located in front and to the left of the esophagus. Then, at the level of the VIII-IX thoracic vertebrae, the aorta bends around the esophagus on the left and goes to its posterior surface. To the right of the thoracic part of the aorta are the azygos vein and the thoracic duct, to the left is the parietal pleura adjacent to it, at the point where it passes into the posterior part of the left mediastinal pleura. In the chest cavity, the thoracic part of the aorta gives off paired parietal branches; posterior intercostal arteries, as well as visceral branches to the organs of the posterior mediastinum.
The abdominal part of the aorta, pars abdomindlis aortae,
being a continuation of the thoracic part of the aorta, it begins at the level of the XII thoracic vertebra, passes through the aortic opening of the diaphragm and continues to the level of the middle of the body of the IV lumbar vertebra.
The abdominal part of the aorta is located on the anterior surface of the lumbar vertebral bodies, to the left of the midline; lies retroperitoneally. To the right of the abdominal aorta are the inferior vena cava, anteriorly - the pancreas, the horizontal (lower) part of the duodenum and the root of the mesentery of the small intestine. The abdominal part of the aorta gives off paired parietal branches to the diaphragm and to the walls of the abdominal cavity, and itself directly continues into the thin median sacral artery.
The visceral branches of the abdominal aorta are the celiac trunk, the superior and inferior mesenteric arteries (unpaired branches) and the paired renal, middle adrenal and testicular (ovarian) arteries.
Branches of the aortic arch
Brachiocephalic trunk, truncus brachlocephdlicus,
originates from the aortic arch at level II of the right costal cartilage.
In front of it is the right brachiocephalic vein, behind it is the trachea. Heading upward and to the right, the brachiocephalic trunk does not give off any branches and only at the level of the right sternoclavicular joint is divided into two terminal branches - the right common carotid and right subclavian arteries.
Right common carotid artery, a.
carotis communis dextra, is a branch of the brachiocephalic trunk, and the left common carotid artery,
a.
carotis communis sinistra, arises directly from the aortic arch.
The left common carotid artery is usually 20-25 mm longer than the right one. The common carotid artery lies behind the sternocleidomastoid and scapulohyoid muscles, follows vertically upward in front of the transverse processes of the cervical vertebrae, without giving off branches along the way.
Outward from the common carotid artery are the internal jugular vein and the vagus nerve, inwardly - first the trachea and esophagus, and above - the larynx, pharynx, thyroid and parathyroid glands.
At the level of the upper edge of the thyroid cartilage, each common carotid artery is divided into the external and internal carotid arteries, which have approximately the same diameter. This location is called the bifurcation of the common carotid artery. A small expansion at the beginning of the external carotid artery is the carotid sinus, sinus caroticus.
In the area of the bifurcation of the common carotid artery there is a small body 2.5 mm long and 1.5 mm thick - the carotid glomus,
glomus caroticum
(carotid gland, intercarotid glomerulus), containing a dense capillary network and many nerve endings (chemoreceptors).
External carotid artery, a.
carotis externa, is one of the two terminal branches of the common carotid artery. It is separated from the common carotid artery within the carotid triangle at the level of the upper edge of the thyroid cartilage. Initially, it is located medial to the internal carotid artery, and then lateral to it. The initial part of the external carotid artery is externally covered by the sternocleidomastoid muscle, and in the area of the carotid triangle - by the superficial plate of the cervical fascia and the subcutaneous muscle of the neck.
Located medially from the stylohyoid muscle and the posterior belly of the digastric muscle, the external carotid artery at the level of the neck of the mandible (in the thickness of the parotid gland) is divided into its final branches - the superficial temporal and maxillary arteries. On its way, the external carotid artery gives off a number of branches that extend from it in several directions.
The anterior group of branches consists of the superior thyroid, lingual and facial arteries. The posterior group includes the sternocleidomastoid, occipital and posterior auricular arteries.
The ascending pharyngeal artery is directed medially.
Anterior branches of the external carotid artery
Superior thyroid artery, a. thyreoidea superior
Lingual artery, a. lingualis,
Facial artery, a. facidlis,
Posterior branches of the external carotid artery:
1. Occipital artery, a.
occipitdlis
2. Posterior auricular artery, a. auriculdris posterior
The medial branch of the external carotid artery is the ascending pharyngeal artery, a.
pharyngea ascendens. This is a relatively thin vessel, departs from the internal semicircle of the external carotid artery at its beginning, and rises up to the lateral wall of the pharynx.
From the ascending pharyngeal artery depart: 1) pharyngeal branches, rr. pharyngedles,
to the muscles of the pharynx and to the deep muscles of the neck;
2) posterior meningeal artery, a.
meningea posterior, follows into the cranial cavity through the jugular foramen;
3) inferior tympanic artery, a.
tympdnica inferior, penetrates into the tympanic cavity through the lower opening of the tympanic canaliculus.
Terminal branches of the external carotid artery
Superficial temporal artery, a. tempordlis superficid-lis,
Maxillary artery, a. maxilldris,
Endovascular interventions
They allow you to dramatically reduce the amount of surgical trauma, shorten hospitalization periods and reduce the inevitable suffering of the patient associated with surgical approaches. One of the main disadvantages of the method is the need for repeated interventions.
Types of endovascular operations for aortic aneurysm:
- implantation of a stent graft into the abdominal aorta,
- implantation of a stent graft into the ascending (thoracic) aorta.
The most modern method of treating aortic aneurysm is a hybrid method, which allows achieving optimal treatment results with the least surgical trauma.
Hybrid surgeries combine the advantages of open and endovascular interventions.
To prevent the development of aortic aneurysms, the most important thing is the need to control risk factors, namely arterial hypertension. In addition to arterial hypertension, the most significant risk factors are age (over 55 years), male gender, smoking, the presence of aneurysms in direct relatives, and high cholesterol levels.
Functions
The aorta of the heart has a huge role to play.
Being the beginning and main highway of the systemic circulation, it delivers blood to all organs of the chest, abdominal cavity and small pelvis. From its arch originate the vessels that supply blood to the head and upper limbs, and from the abdominal part of the aorta blood enters the lower limbs. From this we can conclude that almost all organs and tissues of the human body are supplied with blood through the aorta of the heart. To the point where the aortic bifurcation begins, arteries branch off from it to all vital organs:
- in the chest cavity - to the heart and surrounding tissues;
- in the abdominal cavity from the infrarenal abdominal aorta - to the gastrointestinal tract and urinary organs;
- in the abdominal cavity from the intrarenal part of the vessel to the organs of the reproductive system.
The abdominal part of the aorta is completed by a bifurcation, that is, the bifurcation of the aorta, into the left and right iliac arteries, through which blood flows to the lower extremities.
In addition to nourishing tissues, the vessel takes part in the regulation of blood pressure. The mechanism of stretching and relaxing the walls prevents the formation of a large gap between blood pressure in systole and diastole.