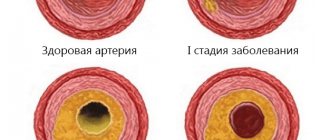
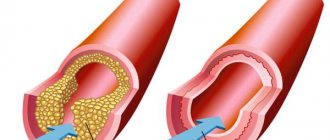
The main cause of death in developed countries is myocardial infarction, which is caused by atherosclerosis of the coronary arteries. Coronary atherosclerosis is manifested by the appearance of atherosclerotic plaques - lesions of the vessel wall, which are rounded convex formations, filled first with almost liquid cholesterol in the form of a foamy substance, and then. gradually scarring (overgrown with connective tissue) and accumulating calcium.
The detection of calcium in the coronary arteries clearly indicates the presence of an atherosclerotic process and atherosclerotic plaques, and the amount of this calcium ( coronary calcium index or Agatston index ) indicates the severity of atherosclerotic lesions.
How is calcium detected in the coronary arteries?
The presence and amount of calcium in the coronary arteries can be determined using cardiac computed tomography (CT). This method is based on sequential X-ray images of the human body at different levels, which are then combined by a computer into a complete picture. Modern computed tomographs are capable of creating a three-dimensional image of the organ being examined, including blood vessels.
CT scan of the coronary arteries to determine calcium levels is a non-invasive technique (performed without penetration into the internal environment of the body).
CT image. The white areas indicated by arrows are calcium deposits in the coronary arteries.
Material and methods
An analysis of the literature was carried out in the search engines Pubmed, GoogleScholar, Scopus and RSCI with a list of keywords “coronary artery calcification”, “coronary artery calcification score”, “coronary artery calcification”, “calcium index”.
This review included 51 papers from studies conducted from 1990 to 2021 that described the use of various scales to assess the severity of coronary artery disease, as well as studies that examined the results of direct myocardial revascularization for coronary artery calcification. Pathogenesis of coronary artery calcification
For a long time, it was believed that the mechanisms of development of CCA represent a passive degenerative process and some final stage of atherosclerosis, which was confirmed by the correlation of the degree of calcification with the age of the patient [5]. Modern researchers are inclined to believe that arterial calcification is an active process, which is based on mechanisms regulating calcium metabolism, in particular the mechanisms of growth and bone formation [6]. There is a concept that considers atherosclerosis as a chronic inflammatory process that induces osteogenic differentiation of vascular smooth muscle cells (VSMCs), which leads to CCA [7]. There is no doubt that plaque calcification begins already at the stage of formation of lipid bands and only progresses at all other stages of atherogenesis. It is currently believed that the mechanism of coronary artery calcification is similar to the process of bone tissue formation.
There are two recognized morphological types of CCA: atherosclerotic calcification with predominant damage to the intima and calcification of the medial layer of the arteries. In the first type, osteogenic differentiation of VSMCs is induced by inflammatory mediators and lipids of atherosclerotic plaques [7]. The second scenario is associated with advanced age, diabetes, and chronic kidney disease (CKD). Previously considered a benign process, medial calcification contributes to increased arterial stiffness, which increases the risk of adverse cardiovascular events [8]. Calcification of the coronary arteries in both variants leads to a decrease in the elasticity of the artery wall, pathological vasomotor responses and impaired myocardial perfusion [9].
It is known that a plaque with a calcified cap is much more stable and resistant to rupture than a “soft” plaque and even a normal vascular wall [10]. Apparently, this conclusion is applicable only in the case of homogeneous calcification. According to some studies, in patients with acute coronary syndrome, multiple small calcium inclusions, called “motley” or “spotty”, are detected, while in chronic coronary artery disease larger and uniform calcifications are determined [11]. The zone formed between the calcified cap and the non-calcified vascular wall is considered to be a potential rupture zone [12]. When performing PCI, there is a high probability of developing dissection in such a zone; Large plaques with obvious “patchy” calcification have been described to have a tendency to rupture [13].
Osteopontin, osteoprotegerin, RANKL, fetuin-A, and bone morphogenetic proteins play a role in the development of calcification. All these substances are produced in the vascular wall during the progression of atherosclerosis; their participation in the regulation of plaque calcification has been proven. A number of studies have revealed a relationship between the level of osteopontin and the level of coronary calcium measured using multislice computed tomography (MSCT) [14]; It was proposed to consider osteopontin as an independent risk factor for cardiovascular events. It has been shown that osteopontin and bone morphogenetic protein type 7 determine the differentiation of VSMCs into osteoblast-like cells and induce calcium deposition in the vascular wall, and osteoprotegerin plays an inhibitory role in vascular calcification [15-17]. Normally, there is a balance between the regulators of calcification, but CCA can develop when the balance is disturbed in favor of the inducers. The exact mechanisms of this process remain to be studied.
Prevalence of coronary artery calcification
The prevalence of CCA depends on age and gender. According to most authors, in the age group over 70 years, CCA occurs in more than 90% of men and more than 67% of women [18]. A high risk of developing CCA is observed in patients with high body mass index, high blood pressure, dyslipidemia, hyperglycemia, family history, CKD, high levels of fibrinogen and elevated levels of C-reactive protein [19], i.e., with all generally recognized risk factors for atherosclerosis .
Diagnostics
Computed tomography (CT) is the mainstay of noninvasive diagnosis of CCA; The method is able to quantify calcification and has high sensitivity and specificity. Multislice computed tomography (MSCT) is based on the measurement and computer processing of the difference in attenuation of X-ray radiation by tissue density. To quantify the degree of CCA, a calculated indicator is used - the calcium index (CI). CI correlates with the severity of coronary atherosclerosis, the presence of hemodynamically significant coronary artery stenoses and the risk of developing coronary complications [20]. The CI calculation is used according to the method proposed in 1990 by A. Agatston et al. [21]: CI is calculated by multiplying the area of calcified lesion of the coronary artery by the conditional density factor. The density factor is calculated from the peak density of the calcified zone, expressed in Hounsfield units (G. Hounsfield) - HU. It is taken as 1 unit. for calcifications with a density of 130-199 HU, for 2 units. — for calcifications with a density of 200-299 HU, for 3 units. - for calcifications with a density of 300-399 HU and for 4 units. - for calcifications with a density of 400 HU or more. So, for example, when detecting calcification with an area of 6 mm2 with a peak density of 265 HU, the CI will be 12 units. (6×2), and for calcification of the same area, but with a peak density of 432 HU - already 24 units. (6x4). Total K.I. is calculated as the sum of the indices determined on all tomographic slices. Algorithms for calculating volumetric CI and calculating the mass of calcium phosphate have also been proposed [22]. The American College of Cardiology and the American Heart Association (2010) consider it useful to non-invasively measure the degree of CAC to assess the risk of cardiovascular disease in asymptomatic patients with intermediate risk (10-year risk 10-20%); recommendation class IIa [23].
It has been shown that as the calcium index increases, sensitivity is lost and specificity in predicting coronary artery disease increases [24]. In other words, in severe coronary artery calcification and very high CI, it becomes difficult to detail the topography of the lesion and its extent. Based on this, Z. Qian et al. proposed separate methods for assessing calcification of atherosclerotic plaques (lesion-specificscore) and coronary arteries (vessel-specificscore) as an addition to the already existing Agatston scale. The use of lesion-specific and vessel-specific calcium score increases the sensitivity of the study (with 80% specificity), which is superior to the traditional Agatston score in predicting IHD [25].
Coronary angiography has lower sensitivity compared to CT scanning in determining CCA, but at the same time has high specificity. According to angiography, CCA is assessed using the following criteria: 1) assessment of calcification of the target vessel on a 4-point scale (0 - no calcification, 1 - barely noticeable calcification, 2 - easily visualized, moderate calcification and 3 - severe calcification), 2) depth calcification after administration of contrast (superficial with calcification closer to the lumen of the vessel, deep with calcification closer to the adventitia), 3) is CCA determined reliably in two or more orthogonal projections and 4) is CCA determined in areas other than the target vessel [26] .
Intravascular ultrasound (IVUS) is a more accurate method for diagnosing coronary arteries than angiography, with high sensitivity (90-100%) and specificity (99-100%). A calcified plaque on IVUS appears as an echogenic shadow with acoustic shadowing, and the degree of calcification can be assessed by several indicators. According to the range of calcified lesions on IVUS, 4 classes are distinguished: class 1 (calcified lesion angle from 0 to 90°), class 2 (CAC angle from 91 to 180°), class 3 (CAC angle from 181 to 270°) and grade 4 (KKA angle from 271 to 360°). The location of calcium is defined as superficial (present in the intimal layer), deep (present in the medial adventitial layer) and mixed. Calcium deposits are assessed in the thickest atherosclerotic plaque [27].
Optical coherence tomography (OCT) is the optical analogue of intravascular ultrasound; it also has high sensitivity and specificity for identifying CCA. The difference in the physical principle of operation of these two methods is that with OCT, not an acoustic wave is used to study biological tissues, but infrared light radiation with a wavelength of about 1300 nm. However, the resolution of OCT (up to 10-20 µm) is approximately 10 times higher than that of IVUS (up to 100-150 µm), which makes it possible to differentiate the intima, media and adventitia. H. Yabushita et al. [28] in their analysis of OCT data described the specific features of each type of atherosclerotic plaque: fibrous plaque is characterized by a homogeneous area of high signal with low attenuation, calcified plaque is characterized by a well-defined area with low signal and clear boundaries, and lipid-rich plaque is characterized by an area of low signal and diffuse boundaries. . Despite its high resolution, OCT has a number of limitations that can pose a challenge in measuring the area of calcification and visualizing deep vascular structures. Thus, the maximum depth of signal penetration is 1-2 mm (for IVUS - up to 4-8 mm), and absorption by hemoglobin and scattering on erythrocytes lead to strong attenuation of the signal [29].
Thus, clinicians today have diagnostic tools at their disposal that allow them to assess coronary artery calcification both qualitatively and quantitatively. However, it must be recognized that convenient and non-invasive methods are more suitable for screening for coronary disease. For a detailed assessment, including the extent of calcification and involvement of the distal segments of the artery, an expensive invasive technique is required and, possibly, a comparison of its data with data obtained intraoperatively.
Percutaneous coronary intervention
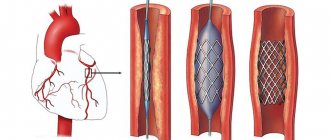
Coronary calcification increases the likelihood of developing complications of angioplasty and is therefore often the reason for refusing to perform it [30]. The pressure exerted on the vessel wall when the balloon is inflated may be uneven due to varying degrees of calcification; this increases the risk of dissection, acute vessel occlusion, the possibility of subsequent restenosis and the development of adverse cardiovascular events [31]. Severe CCA creates difficulties during device delivery and increases the risk of vessel embolization, which in turn leads to an increase in the incidence of periprocedural MI [32].
With the introduction of bare metal stents (BMS), early and long-term survival has improved. However, incomplete stent deployment, asymmetric deployment, incorrect placement, or stent migration, observed with severe CCA, increased the risk of stent restenosis and thrombosis [33].
The use of drug-eluting stents (DES) has proven to be more effective. According to the TAXUS-IV study, in patients with calcified lesions, the rate of target vessel ischemia at 1 year was 56% lower with DES compared with BMS (5.1% vs. 11.9%, p
=0.09), however, in patients with non-calcified coronary arteries this difference was significantly greater (75% lower and 4.3% versus 15.7%,
p
<0.0001) [3].
Similar results were described in a meta-analysis by B. Zhang et al. (2015): the use of DES significantly reduces the need for repeated revascularization of target vessels compared with BMS in patients with CCA (8.5% versus 16.0%; relative risk: 0.50; 95% confidence interval: 0.38–0. 65; p
<0.00001) [34]. However, there are also studies reporting similar rates of thrombosis and restenosis of the DES and GMS in patients with CCA, with comparable rates of mortality and MI [2, 3].
Thus, a sequential study of the results of endovascular treatment of patients with coronary artery disease with calcified coronary arteries showed that the best results were obtained with implantation of drug-eluting stents. On the other hand, the results of stenting were compared with those in patients without calcification. They indicate a higher rate of restenosis and repeat revascularization in patients with CCA [35].
Potential risk factors for restenosis and repeat revascularization, such as incomplete stent deployment, damage to the drug coating of the stent due to CCA, and the use of other devices (including rotational atherectomy), may directly contribute to neointimal hyperplasia [36].
Cutting and notching balloon catheters do not remove calcium, but improve the elasticity of arterial walls by creating discrete incisions in the atherosclerotic plaque, which allows increasing the area of work on the affected parts of the artery and reducing the narrowing of the stent, preventing vessel dissection. The indication for a cutting balloon is a relatively short lesion (<20 mm). For prolonged and circular lesions, the use of such balloons is not recommended. In addition, the pressure in the cutting cylinder should not exceed 12 atm to avoid cutting the cylinder blade into the vessel wall [37].
Rotational atherectomy, unlike a cutting balloon, excises hard coronary calcium tissue to produce small particles (<10 µm) without affecting soft elastic tissue. Patients with CCA undergoing rotational atherectomy have an increased risk of thrombosis, development of the no-reflow phenomenon with an increased risk of periprocedural MI [38]. However, the use of rotational atherectomy has been found to be clinically effective in patients with CCA [39]. In order to improve the prognosis after exposure, implantation of a DES is recommended. There are a number of studies reporting favorable long-term results after DES implantation followed by rotational atherectomy [40].
Laser coronary atherectomy uses pulsed excimer or holmium laser energy to generate high-energy transient waves; There is a photoacoustic effect on resistant atherosclerotic lesions. Despite the fact that the method was introduced more than two decades ago, due to its uncertain results, as well as due to the advent of DES, laser angioplasty has lost its practical significance as an independent intervention and its use is limited to a few centers. Some studies have demonstrated potential procedural complications such as vessel dissection (especially vessels with superficial calcium), perforation, and a high risk of restenosis [41]. However, the procedure can be used in patients with CCA to destroy calcium before stent implantation in cases where there is a risk of incomplete stent deployment [42].
Orbital atherectomy, like rotational atherectomy, has a differential ablative effect on hard and soft surfaces, producing particles <2 μm in size when centrifugal force is applied to the vessel wall. The device allows operators to control the ablation depth by increasing the rotation speed (from 60,000 to 120,000 rpm). Like rotational atherectomy, orbital atherectomy improves the elasticity of arterial walls to reduce procedural complications and facilitate stent implantation. According to J. Chambers et al., the use of orbital atherectomy for severe coronary calcification not only improved stent delivery, but also improved early and 30-day clinical outcomes compared with the results of previous studies in a similar cohort of patients [43].
Thus, the evolution of endovascular revascularization and analysis of its results allow us to look optimistically at the prospects for treating patients with coronary artery disease, however, the study of immediate and delayed results shows less effectiveness of treatment if the coronary arteries are calcified.
Coronary artery bypass surgery
The fact that coronary artery calcification is a predictor of a worse prognosis after PCI leads clinicians to consider surgical revascularization as a priority treatment option in this situation. However, the question of the prognostic value of CCA for patients undergoing CABG remains unclear, and the available data do not allow us to draw solid conclusions. There are only a few studies aimed at addressing this issue. Of interest is the work of M. Castagna et al., who expressed a judgment about the more frequent development of calcification of autovenous shunts in patients with initial calcification of native coronary arteries [44].
In the analysis of K. Ertelt et al. reported on 755 patients with ACS who were included in the ACUITY study (Acute Catheterization and Urgent Intervention Triage Strategy Trial) with a follow-up period of 1 year after CABG [45]. The authors found that severe calcified lesions of the coronary artery were an independent predictor of major adverse cardiovascular events: when comparing 1-year mortality in patients with severe ( n
=103), moderate (
n
=249) and absent calcification (
n
=403) it was 11.8, 3.7 and 4.5%, respectively,
p
=0.006.
In a similar study by C. Bourantas et al. (2015) included 1545 patients (896 from the SYNTAX registry and 645 from the SYNTAX CABG registry) for 5 years of follow-up after CABG. Patients with severe calcification were compared ( n
=548) and without significant calcification of the coronary arteries (
n
=997).
Patients with severe CCA had a higher mortality rate: 17.1% versus 9.9%, p
<0.001, but the incidence of adverse nonfatal cardiovascular events was similar in the groups (26.8% versus 21.8%,
p
= 0.057 ). The higher mortality in the severe calcification group was partly explained by the presence of more severe comorbidities (renal failure, hypertension) and multifocal atherosclerosis [1]. A significant limitation of the study is the fact that the characterization of calcification was based on angiographic data without the use of MSCT or IVUS.
Drug treatment
To date, there is no generally accepted conservative treatment for CCA. The role of statins in the treatment of patients with CCA is unclear. According to numerous studies, statin therapy does not have a significant effect on arterial CCA [46]. Some researchers even express the opinion that statins can enhance the process of calcification [47]. Non-randomized studies have shown regression of CCA with the use of calcium channel blockers, hormonal therapy, and phosphate binders [48, 49].
In drug therapy, the assessment of the effect of taking calcium supplements deserves special mention. Based on the results of the large EPIC-Heidelberg study, which included 24 thousand people aged 35 to 64 years, the authors argue that the use of calcium supplements can significantly increase the risk of developing myocardial infarction [50]. According to some data, the risk of myocardial infarction increases with the use of calcium supplements at a dose of more than 800 mg/day [7]. There are also studies that have led to the opposite conclusion: dietary calcium does not have a significant effect on vascular calcification and cardiovascular events [51].
When is a cardiac CT scan prescribed to study the calcium index of the coronary arteries?
The purpose of the study is to detect the presence of coronary atherosclerosis, determine its severity and use the data obtained to predict the course of the disease. Thus, the study is prescribed to patients with suspected coronary heart disease, even in cases where there are no clinical symptoms. The most relevant determination of the calcium index of the coronary arteries is for persons with risk factors:
- high blood cholesterol levels,
- history of heart attacks in close relatives,
- diabetes,
- high blood pressure,
- smoking,
- obesity,
- low physical activity (physical inactivity).
Coronary calcium indexing
Calcification of arterial walls reflects the development of atherosclerosis and does not occur in normal arteries. Indexation of coronary calcium as an indicator of atherosclerosis of the coronary arteries began to be used in medical practice relatively recently.
Because CT is very sensitive to detecting high-density structures, numerous methods have been proposed for detecting and quantifying coronary calcium, ranging from single-slice scans without ECG-synchronization to ECG-gated multislice scans and ECG-triggered electron beam scans.
Various automatic index calculation programs are available in clinical practice. The user must identify potential plaques, assign them to different parts of the coronary arterial tree, and place R01 around them. Further assessment is carried out automatically through the program. As a rule, a threshold value of 130 units is used. X, but for assessments that are not based on EBCT data, a threshold of 90 units has been proposed. X.
Multislice CT scans synchronized with ECG should be the standard when assessing coronary calcium by multislice CT. The obtained data are assessed either by the Agatston index or by measuring the total mass of coronary calcium. The Agatston index is a traditional measure of coronary calcium using a cutoff value of 130 units. X to identify the calcified portions of the coronary arteries (Fig. 6).
The total area of each calcified area (over 1 mm2, or 1–3 pixels in size) is weighted by a factor based on the peak density in that area and summed. The sum of the individual indices constitutes the total calcium index (Agatston index). Risk classification is based on the Agatston index and is presented in table. 3.
New methods include determining the total volume of calcified plaques, or the total mass of coronary calcium (calculated from the volume of plaques and their average density). Total calcium mass can be individually calibrated using a calcium hydroxyapatite phantom, which is placed on the bed underneath the patient during scanning. Total calcium mass has the greatest reproducibility because it is less sensitive to partial volumetric effects than other coronary calcium measures.
The calcium volume score (CVS) was proposed by Callister (1998) to improve reproducibility compared to the Agatston index. This uses interpolation between slices and determines the volume of calcified plaques. CVS has been used successfully to demonstrate regression of calcified plaques in patients treated with hydroxymethylglutaryl-coenzyme A inhibitors.
But it should be remembered that although the amount of coronary calcium is in close relation to the total plaque mass, calcification represents only the tip of the iceberg. The determined volume of calcified plaques is 20% of the total plaque mass. There may even be some patients without calcification but with easily vulnerable plaques that are prone to rupture.
How to prepare for a coronary CT scan?
No special preparation for the study is required. You must continue taking your prescribed medications and avoid drinking coffee and smoking for 4 hours before the test.
The quality of computed tomography of the coronary arteries can be affected by jewelry (including piercings) and metal hair clips, so it is advisable to leave them at home. The person being examined will also be asked to remove their hearing aids and removable dentures.
3.How is screening carried out?
A CT scan of the body is performed by a technologist and interpreted by a radiologist. Other doctors may also use the resulting images.
You will be asked to remove all jewelry and undress. You will be given a diaper to cover yourself during the procedure. You will need to lie down on a table to which the CT scanner is attached.
The table then slides into the CT scanner and it moves around your body to take pictures. You may hear noises during the procedure. It is important to remain still during the procedure.
If necessary, they can perform myelography, i.e. CT scan using a special dye. The contrast agent is usually injected into a vein in the arm. After this, wait some time for the dye to spread throughout the body.
The procedure takes from 30 to 60 minutes.
How to prepare for coronary calcium screening?
Before a coronary calcium screening, tell your doctor if you may or may be pregnant. You may be asked not to eat or drink anything containing caffeine for a while before the test.
About our clinic Chistye Prudy metro station Medintercom page!
How is the cardiac CT procedure performed to study the calcium index of the coronary arteries?
During the examination, the patient lies on his back on a special table. Electrodes are attached to his body to simultaneously record an electrocardiogram. This is necessary so that the CT scanner takes pictures only in the intervals between heartbeats. after placing the patient, the table moves quickly until it reaches the position required to begin the study, and then slowly moves allowing the device to take pictures of different parts of the body (in this case, parts of the heart). CT scan of the coronary arteries does not cause any sensations.
During the examination, the patient may be asked to hold their breath for 10-20 seconds.
The total duration of the study is about 10 minutes. The research physician is not in the room during the study, but is always available for contact via a communication device.
Artifacts
1. When evaluating calcium index data, care must be taken not to include calcifications of the aortic bulb at the openings of the right and left coronary arteries. Mitral valve calcifications should not be mistaken for LOA calcifications.
2. Artifacts from motion may be acceptable in determining coronary calcium, but interfere with follow-up if high reproducibility is required.
3. Image noise reduces the accuracy of detection and quantification of coronary calcium and reduces the reproducibility of such measurements.
4. Pulsatility artifacts can be used to determine the movement of cardiac structures or major vessels. But pulsation is the main reason for uninformative studies of the coronary arteries.
5. Arrhythmia is the main cause of suboptimal results in either prospectively triggered EBCT or MCT. Retrospective synchronization can compensate for changes in heart rate, but extrasystoles may be accompanied by incomplete or pathological contraction of the heart muscle, which will cause non-synchronization with the rest of the data set. Arrhythmia may cause pseudostenoses of the coronary arteries or hide stenoses that would otherwise remain within the scanned volume. Therefore, this method should not be attempted in such patients.
6. Artifacts caused by respiratory movements are eliminated by training and education of the patient.
7. Artifacts caused by high contrast (streak-like artifacts).
7. Personal volumetric effect.
8. Superposition of structures (difficulties in differentiating structures).
How much does a coronary CT scan with calcium index cost?
We called all specialized medical centers performing computed tomography in Moscow, which Yandex showed on the first page of the search. In most of them, administrators heard the name of the method for the first time. However, we found one clinic in which CT of the coronary arteries with determination of the calcium index is still performed. The price of the study is from 3,500 rubles (data as of November 2021) We hope that in the near future the study of the calcium index of the coronary arteries will become available in all computed tomography centers.
| In the near future, it will be possible to accurately determine the risk of cardiovascular accidents through a simple blood test. |
Our comment.
The study of the calcium coronary index is an important tool that allows you to clarify the risk of cardiovascular disease and individualize treatment and preventive approaches. It is advisable to use calcium index data together with risk determination using the SCORE scale.
Discuss the material or ask questions on Facebook.
Coronary CT angiography
Coronary CT angiography (CTA) offers excellent opportunities for non-invasive visualization of the coronary vessels [7, 11, 14, 16–18, 22–24] (Fig. 4, 5).
Coronary CT angiography using electron beam scanning and a trigger signal corresponding to 40–80% of the RR interval (depending on the patient's heart rate) can cover the entire heart. Single-layer mode is used with a rotation time of 100 ms. Adequate training of patients is necessary to ensure that they can hold their breath for 40–50 heart beats.
Triphasic electron beam angiography was developed to optimize the imaging of the coronary arteries in patients with arrhythmia. This technology collects data for three layers during each cardiac cycle (minimum time difference between phases is 58 ms).
For multi-slice CT scanning, retrospective synchronization with the ECG is required. Multisector reconstruction provides better results but requires more radiation exposure to the patient.
Helps show the external shape of the heart chambers and great vessels and display the anatomy of the coronary arteries with volumetric representation (VRT). With VRT it is often easier to recognize the different branches of the coronary arteries than with transverse sections. Some manufacturers offer presets that allow selective display of the heart (excluding the chest wall and pulmonary vascularity) and even the coronary arterial tree.
When performing multislice computed tomography with intravenous contrast, it is possible to visualize the coronary arteries over a sufficient length. At the same time, 78 ± 16% of arterial segments were visualized without artifacts [10]. The sensitivity and specificity of multislice computed tomography in detecting significant stenoses (more than 50%) of the coronary arteries are quite high - 90 and 85%, respectively [18, 19]. Based on this, multislice computed tomography, given its non-invasiveness and relative ease of performance, can compete with coronary angiography, but not replace it. EBCT and MCT-based CTA have been found to provide a sensitivity of 60 to 90% in the diagnosis of coronary stenosis, depending on the caliber of the affected vessels [18]. Proximal stenoses are detected more accurately than distal or small artery stenoses.
It should be remembered that virtual angioscopy of the coronary arteries is a method that cannot be used to evaluate the complex nature of the branching or aneurysms of the coronary arteries [5]. In addition, virtual angioscopic images are subject to blooming effects, which make calcified plaques appear much larger than in reality. Suspicious areas must be analyzed in at least two mutually perpendicular planes. The suspected stenosis must then be classified and assigned to a specific segment of the coronary tree. In addition, it is necessary to determine the nature of the stenosis, taking into account the composition of the plaque. Blooming of calcified plaques creates difficulties that can be reduced by using a wide window setting for image analysis. Extensive calcified plaques are characterized by longer segments of irregular calcification.
The thickness of the normal arterial wall is 0.1 mm and is therefore not visible on CTA. Calcified, or hard, plaques have a significantly higher density than fibrous tissue (90 units X) (Table 2).
Calcified nodules are small pinpoint inclusions of calcium surrounded by compacted soft tissue. They are rarely accompanied by acute cardiac attacks and are considered as atheromas that do not rupture, but are completely calcified. Widespread calcification of the coronary arteries without non-calcifying plaques rarely reflects the location of significant stenosis.
Fibrous-calcified plaques on coronary CTA are characterized by longer segments of irregular calcification combined with dense fibrous tissue (approximately 90 X units). They are considered as a consequence of plaque rupture with negative restructuring, causing the proliferation of scar tissue and stenosis. Such plaques and their accompanying stenosis are particularly difficult to evaluate by CTA because of the blooming effect, which causes the calcifications to appear larger than they actually are (due to limitations in spatial resolution).
Soft plaques do not contain calcifications and can be preatheromas, atheromas, fibroatheromas, and fibrous plaques. Preatheromas are thin (1 mm), layers of soft plaques inseparable from the wall. They are rarely detected on CTA. Atheromas contain a large lipid (cholesterol) core and are described as having a density of 50 units. X or lower. On CTA, the fibrous coating is not differentiated from the lipid core. Atheromas are adherent to the wall, may be localized, or appear as a long area of irregular, hypodense wall thickening. Remodeling may cause the lumen to appear unchanged despite significant plaque burden, but atheromatous lesions may also cause significant stenosis even in patients with a calcium score of 0.
Fibrous plaques are denser, their CT density is approximately 90 units. X. They are often accompanied by stenosis. Fibroatheromas have an intermediate CT density from 50 to 90 units. X.
Coronary thrombi can be suspected if the artery has a dark center and a light ring-shaped border.
CT angiography is not sensitive enough to accurately determine the degree of coronary artery stenosis due to the small caliber of these vessels and significant difficulties caused by pulsatile effects. Therefore, a scheme for assessing the degree of stenosis using five categories has been proposed:
— 0 (0%) — normal;
— I (1–49%) — non-obstructive lesion;
— II (50–74%) — significant stenosis;
— III (75–99%) — high degree stenosis;
— IV (100%) — occlusion.
Cardiac ischemia
In patients with chronic ischemia, CT can reveal scarring, thinning of the ventricular wall, dyskinetic wall segments, and aneurysms. These changes can be detected already with conventional (not synchronized with ECG) CT, but are often not assessed. Although there have been attempts to study perfusion using CT aimed at determining myocardial viability, scintigraphy and MRI are currently more suitable for this task [1, 5, 8].
CT morphology
Acute infarction leads to ischemia, myocardial damage, and ultimately scar formation. On delayed scanning (10 to 40 minutes after contrast agent administration), the damaged tissue should become hyperdense due to increased retention of contrast agent in the interstitium.
The formation of subendocardial scars can be assessed by a ring-shaped zone of hypodensity in the endocardial layer of the ventricles. Subendocardial, but more often transmural infarctions lead to local thinning of the ventricular wall with regional disturbance of wall motion. As a result, aneurysms and blood clots may form.
Different parts of the myocardium can be attributed to certain basins of the coronary vessels, but this is not reliable enough in the area of the base of the heart and depends on which system the posterior descending coronary artery belongs to (right coronary artery, left coronary artery).
CT morphology of cardiac aneurysms. The wall of true aneurysms has the appearance of a protrusion of the contour of the heart, local thinning is noticeable, and a characteristic paradoxical movement in systole is detected. Occasionally, ring-shaped calcification is observed in the fibrous wall. There is always a wide connection with the heart chamber (there is no neck).
Cardiac pseudoaneurysms may have a spherical shape and a neck that is smaller than the true diameter of the aneurysm. They are usually located along the posterolateral and diaphragmatic walls of the left ventricle. Due to the delay in filling of the aneurysm, the intensity of its contrast may differ from the intensity of the lumen of the ventricle.
Echocardiography is preferable to visualize cardiac chamber thrombosis. CT is superior in identifying thrombi in areas difficult to reach by transthoracic echocardiography.
Coronary venous bypass grafting
Coronary artery bypass grafting (CABG) is usually performed in the middle third of the ascending aorta, although superior or inferior anastomoses (eg, to the brachiocephalic trunk) are also possible. Left coronary artery shunts are placed over the pulmonary trunk.
Evaluation of internal mammary artery grafts using the left or right artery is difficult due to the multiple clips used to occlude small side branches.
The role of CT is to show the patency of the shunts. This does not require synchronization with the ECG. Assessing vein graft stenoses during coronary artery bypass grafting is possible, but very difficult, even using EBCT and multislice-gated CT.
CT morphology
Since the diameter of the venous shunt is usually 3–7 mm, AVBG is usually visualized on CT. The shunt is not contrasted when occluded, whereas stenotic shunts remain fully contrasted. Stenoses most often occur at the site of anastomosis. Over time, calcifications may form in the shunt wall without causing occlusion or stenosis. A narrow (1–5 mm wide) strip of soft tissue density along the shunt means chronic occlusion.
With shunts with transplantation of the right or left internal thoracic arteries, patency is usually detected, but their lumen is very small (1–4 mm). It is not yet possible to reliably recognize stenosis.